中文摘要:
对于心血管疾病(CVD)的手术治疗,显然需要且有待开发小直径(直径<6 mm)的血管移植物。在我们先前的工作中,由于具有很好的细胞相容性和血液相容性,硫酸化丝素蛋白(SF)被成功地制造作为潜在的制备血管移植物的材料。然而,单层血管移植物难以适应复杂的内部环境。在本研究中,聚己内酯(PCL)和硫酸化SF被用于制造双层血管移植物(BLVG),以模拟天然血管的结构。为了增强BLVG的生物活性,将尼可地尔(NIC),一种FDA批准的具有多种生物活性的药物,装载到BLVG中,以制造载有NIC的BLVG。评估了载有NIC的BLVG的形态,化学成分和机械性能。结果表明,载有NIC的BLVG的双层结构使移植物具有双相药物释放行为。体外研究表明,载有NIC的BLVG可以显着增加内皮细胞(EC)的增殖,迁移和抗氧化能力。此外,我们发现潜在的生物学机制是PI3K / AKT / eNOS信号通路的激活。总体而言,结果有效地证明了载有NIC的BLVG作为功能性小直径血管移植物,可具有良好的体外性能。
关键词:
双层血管移植物;尼可地尔;硫酸化丝素蛋白;内皮细胞功能;PI3K / AKT / eNOS通路
作者:
Zheng Xing · Chen Zhao · Chunchen Zhang · Yubo Fan · Haifeng Liu
单位:
Beihang University等
引用本文:
Xing, Z., Zhao, C., Zhang, C. et al. Bilayer nicorandil-loaded small-diameter vascular grafts improve endothelial cell function via PI3K/AKT/eNOS pathway. Bio-des. Manuf. 4, 72–86 (2021). https://doi.org/10.1007/s42242-020-00107-2
全文链接(可点击直接下载PDF)
http://www.jzus.zju.edu.cn/oldversion/opentxt.php?doi=10.1007/s42242-020-00107-2
文章选图
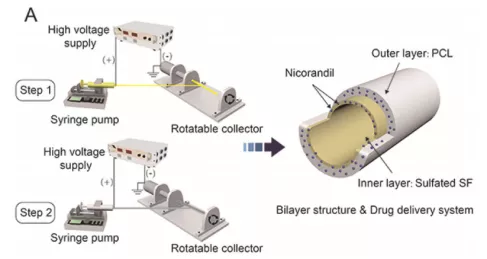
图一 采用两步静电纺丝制备载有NIC的BLVG
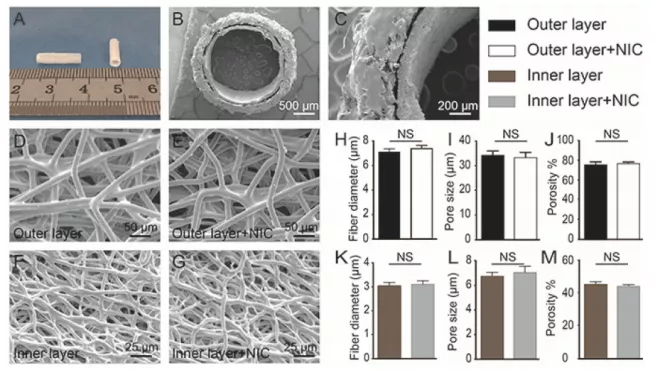
图二 载有NIC的BLVG的形态
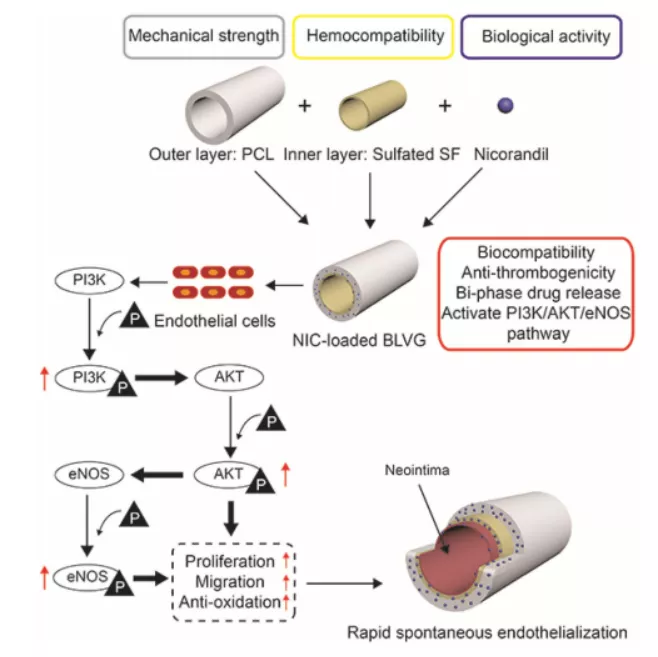
图三 载有NIC的BLVG的生物学功能示意图
参考文献:
1. Ehrmann K, Potzmann P, Dworak C, Bergmeister H, Eilenberg M, Grasl C, Koch T, Schima H, Liska R, Baudis S (2020) Hard block degradable polycarbonate urethanes: promising biomaterials for electrospun vascular prostheses. Biomacromol 21(2):376–387. https://doi.org/10.1021/acs.biomac.9b01255
2. Mathers CD, Loncar D (2006) Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med 3(11):e442. https://doi.org/10.1371/journal.pmed.0030442
3. Bangalore S, Guo Y, Samadashvili Z, Blecker S, Xu J, Hannan EL (2015) Everolimus-eluting stents or bypass surgery for multivessel coronary disease. New Engl J Med 372(13):1213–1222. https://doi.org/10.1161/CIRCINTERVENTIONS.115.002626
4. Wang Z, Mithieux SM, Weiss AS (2019) Fabrication techniques for vascular and vascularized tissue engineering. Adv Healthc Mater 8(19):1900742. https://doi.org/10.1002/adhm.201900742
5. Wang Y, Ma B, Yin A, Zhang B, Luo R, Pan J, Wang Y (2020) Polycaprolactone vascular graft with epigallocatechin gallate embedded sandwiched layer-by-layer functionalization for enhanced antithrombogenicity and anti-inflammation. J Control Release 320:226–238. https://doi.org/10.1016/j.jconrel.2020.01.043
6. Jin X, Geng X, Jia L, Xu Z, Ye L, Gu Y, Zhang AY, Feng ZG (2019) Preparation of small-diameter tissue-engineered vascular grafts electrospun from heparin end-capped PCL and evaluation in a rabbit carotid artery replacement model. Macromol Biosci 19(8):1900114. https://doi.org/10.1002/mabi.201900114
7. Gimbrone Michael A, García-Cardeña G (2016) Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ Res 118(4):620–636. https://doi.org/10.1161/CIRCRESAHA.115.306301
8. Xie RY, Fang XL, Zheng XB, Lv WZ, Li YJ, Ibrahim Rage H, He QL, Zhu WP, Cui TX (2019) Salidroside and FG-4592 ameliorate high glucose-induced glomerular endothelial cells injury via HIF upregulation. Biomed Pharmacother 118:109175. https://doi.org/10.1016/j.biopha.2019.109175
9. Zhang J, Shi J, Ma H, Liu L, He L, Qin C, Zhang D, Guo Y, Gong R (2020) The placental growth factor attenuates intimal hyperplasia in vein grafts by improving endothelial dysfunction. Eur J Pharmacol 868:172856. https://doi.org/10.1016/j.ejphar.2019.172856
10. Chen CC, Hong HJ, Hao WR, Cheng TH, Liu JC, Sung LC (2019) Nicorandil prevents doxorubicin-induced human umbilical vein endothelial cell apoptosis. Eur J Pharmacol 859:172542. https://doi.org/10.1016/j.ejphar.2019.172542
11. Umaru B, Pyriochou A, Kotsikoris V, Papapetropoulos A, Topouzis S (2015) ATP-sensitive potassium channel activation induces angiogenesis in vitro and in vivo. J Pharmacol Exp Ther 354(1):79–87. https://doi.org/10.1124/jpet.114.222000
12. Serizawa K-i, Yogo K, Aizawa K, Tashiro Y, Ishizuka N (2011) Nicorandil prevents endothelial dysfunction due to antioxidative effects via normalisation of NADPH oxidase and nitric oxide synthase in streptozotocin diabetic rats. Cardiovasc Diabetol 10(1):105. https://doi.org/10.1186/1475-2840-10-105
13. Horinaka S, Kobayashi N, Yagi H, Mori Y, Matsuoka H (2006) Nicorandil but not ISDN upregulates endothelial nitric oxide synthase expression, preventing left ventricular remodeling and degradation of cardiac function in dahl salt-sensitive hypertensive rats with congestive heart failure. J Cardiovasc Pharmacol 47(5):629–635. https://doi.org/10.1097/01.fjc.0000211741.47960.c2
14. Joo Myung L, Daiki K, Maki O, Mamoru T, Hiroaki T, Katsuhisa W, Tetsuya A, Akiyoshi K, Hiroki I, Woo-Hyun L, Joon-Hyung D, Chang-Wook N, Nobuhiro T, Bon-Kwon K, Nobukiyo T (2016) Safety and efficacy of intracoronary nicorandil as hyperaemic agent for invasive physiological assessment: a patient-level pooled analysis. EuroIntervention 12(2):208–215. https://doi.org/10.4244/EIJV12I2A34
15. Mao D, Zhu M, Zhang X, Ma R, Yang X, Ke T, Wang L, Li Z, Kong D, Li C (2017) A macroporous heparin-releasing silk fibroin scaffold improves islet transplantation outcome by promoting islet revascularisation and survival. Acta Biomater 59:210–220. https://doi.org/10.1016/j.actbio.2017.06.039
16. Raia NR, Jia D, Ghezzi CE, Muthukumar M, Kaplan DL (2020) Characterization of silk-hyaluronic acid composite hydrogels towards vitreous humor substitutes. Biomaterials 233:119729. https://doi.org/10.1016/j.biomaterials.2019.119729
17. Tozzi L, Laurent PA, Di Buduo CA, Mu X, Massaro A, Bretherton R, Stoppel W, Kaplan DL, Balduini A (2018) Multi-channel silk sponge mimicking bone marrow vascular niche for platelet production. Biomaterials 178:122–133. https://doi.org/10.1016/j.biomaterials.2018.06.018
18. Rodriguez M, Kluge JA, Smoot D, Kluge MA, Schmidt DF, Paetsch CR, Kim PS, Kaplan DL (2020) Fabricating mechanically improved silk-based vascular grafts by solution control of the gel-spinning process. Biomaterials 230:119567. https://doi.org/10.1016/j.biomaterials.2019.119567
19. Gupta P, Lorentz KL, Haskett DG, Cunnane EM, Ramaswamy AK, Weinbaum JS, Vorp DA, Mandal BB (2020) Bioresorbable silk grafts for small diameter vascular tissue engineering applications: in vitro and in vivo functional analysis. Acta Biomater 105:146–158. https://doi.org/10.1016/j.actbio.2020.01.020
20. Li H, Wang Y, Sun X, Tian W, Xu J, Wang J (2019) Steady-state behavior and endothelialization of a silk-based small-caliber scaffold in vivo transplantation. Polymers (Basel) 11(8):1303. https://doi.org/10.3390/polym11081303
21. Liu H, Li X, Zhou G, Fan H, Fan Y (2011) Electrospun sulfated silk fibroin nanofibrous scaffolds for vascular tissue engineering. Biomaterials 32(15):3784–3793. https://doi.org/10.1016/j.biomaterials.2011.02.002
22. Liu H, Li X, Niu X, Zhou G, Li P, Fan Y (2011) Improved hemocompatibility and endothelialization of vascular grafts by covalent immobilization of sulfated silk fibroin on poly(lactic-co-glycolic acid) scaffolds. Biomacromol 12(8):2914–2924. https://doi.org/10.1021/bm200479f
23. Gong X, Liu H, Ding X, Liu M, Li X, Zheng L, Jia X, Zhou G, Zou Y, Li J, Huang X, Fan Y (2014) Physiological pulsatile flow culture conditions to generate functional endothelium on a sulfated silk fibroin nanofibrous scaffold. Biomaterials 35(17):4782–4791. https://doi.org/10.1016/j.biomaterials.2014.02.050
24. Wu T, Zhang J, Wang Y, Li D, Sun B, El-Hamshary H, Yin M, Mo X (2018) Fabrication and preliminary study of a biomimetic tri-layer tubular graft based on fibers and fiber yarns for vascular tissue engineering. Mater Sci Eng C Mater Biol Appl 82:121–129. https://doi.org/10.1016/j.msec.2017.08.072
25. Yin A, Zhuang W, Liu G, Lan X, Tang Z, Deng Y, Wang Y (2020) Performance of PEGylated chitosan and poly (L-lactic acid-co-ε-caprolactone) bilayer vascular grafts in a canine femoral artery model. Colloids Surf B 188:110806. https://doi.org/10.1016/j.colsurfb.2020.110806
26. Yan S, Napiwocki B, Xu Y, Zhang J, Zhang X, Wang X, Crone WC, Li Q, Turng L-S (2020) Wavy small-diameter vascular graft made of eggshell membrane and thermoplastic polyurethane. Mater Sci Eng C Mater Biol Appl 107:110311. https://doi.org/10.1016/j.msec.2019.110311
27. Du H, Tao L, Wang W, Liu D, Zhang Q, Sun P, Yang S, He C (2019) Enhanced biocompatibility of poly(l-lactide-co-epsilon-caprolactone) electrospun vascular grafts via self-assembly modification. Mater Sci Eng C Mater Biol Appl 100:845–854. https://doi.org/10.1016/j.msec.2019.03.063
28. Norouzi SK, Shamloo A (2019) Bilayered heparinized vascular graft fabricated by combining electrospinning and freeze drying methods. Mater Sci Eng C Mater Biol Appl 94:1067–1076. https://doi.org/10.1016/j.msec.2018.10.016
29. Gong W, Lei D, Li S, Huang P, Qi Q, Sun Y, Zhang Y, Wang Z, You Z, Ye X, Zhao Q (2016) Hybrid small-diameter vascular grafts: Anti-expansion effect of electrospun poly ε-caprolactone on heparin-coated decellularized matrices. Biomaterials 76:359–370. https://doi.org/10.1016/j.biomaterials.2015.10.066
30. Seyed S, Zargarian V, Haddadi-Asl Z, Kafrashian M, Azarnia M (2018) Surfactant-assisted-water-exposed versus surfactant-aqueous-solution-exposed electrospinning of novel super hydrophilic polycaprolactone based fibers: analysis of drug release behavior. J Biomed Mater Res Part A 8:675–682. https://doi.org/10.1002/jbm.a.36575
31. Barbara V, Silvia R, Giuseppina S, Maria C, Bonferoni G (2018) Coated electrospun alginate-containing fibers as novel delivery systems for regenerative purposes. Int J Nanomed 10:17–25. https://doi.org/10.2147/IJN.S175069
32. Yang Y, Lei D, Zou H, Huang S, Yang Q, Li S, Qing FL, Ye X, You Z, Zhao Q (2019) Hybrid electrospun rapamycin-loaded small-diameter decellularized vascular grafts effectively inhibit intimal hyperplasia. Acta Biomater 97:321–332. https://doi.org/10.1016/j.actbio.2019.06.037
33. Shi J, Zhang X, Jiang L, Zhang L, Dong Y, Midgley AC, Kong D, Wang S (2019) Regulation of the inflammatory response by vascular grafts modified with Aspirin-Triggered Resolvin D1 promotes blood vessel regeneration. Acta Biomater 97:360–373. https://doi.org/10.1016/j.actbio.2019.07.037
34. Xing Z, Zhang C, Zhao C, Ahmad Z, Li JS, Chang MW (2018) Targeting oxidative stress using tri-needle electrospray engineered Ganoderma lucidum polysaccharide-loaded porous yolk-shell ps. Eur J Pharm Sci 125:64–73. https://doi.org/10.1016/j.ejps.2018.09.016
35. Yao D, Peng G, Qian Z, Niu Y, Liu H, Fan Y (2017) Regulating coupling efficiency of REDV by controlling silk fibroin structure for vascularization. ACS Biomater Sci Eng 3:489–501. https://doi.org/10.1021/acsbiomaterials.7b00553
36. Wang Z, Cui Y, Wang J, Yang X, Wu Y, Wang K, Gao X, Li D, Li Y, Zheng X-L, Zhu Y, Kong D, Zhao Q (2014) The effect of thick fibers and large pores of electrospun poly(ε-caprolactone) vascular grafts on macrophage polarization and arterial regeneration. Biomaterials 35(22):5700–5710. https://doi.org/10.1016/j.biomaterials.2014.03.078
37. Chatterjee S, Judeh ZMA (2015) Encapsulation of fish oil with N-stearoyl O-butylglyceryl chitosan using membrane and ultrasonic emulsification processes. Carbohydr Polym 123:432–442. https://doi.org/10.1016/j.carbpol.2015.01.072
38. Moomand K, Lim LT (2014) Oxidative stability of encapsulated fish oil in electrospun zein fibres. Food Res Int 62:523–532. https://doi.org/10.1016/j.foodres.2014.03.054
39. Singh B, Garg T, Goyal AK, Rath G (2016) Development, optimization, and characterization of polymeric electrospun nanofiber: a new attempt in sublingual delivery of nicorandil for the management of angina pectoris. Artif Cells Nanomed Biotechnol 44(6):1498–1507. https://doi.org/10.3109/21691401.2015.1052472
40. Yao D, Qian Z, Zhou J, Peng G, Zhou G, Liu H, Fan Y (2018) Facile incorporation of REDV into porous silk fibroin scaffolds for enhancing vascularization of thick tissues. Mater Sci Eng C Mater Biol Appl 93:96–105. https://doi.org/10.1016/j.msec.2018.07.062
41. Wei Y, Wu Y, Zhao R, Zhang K, Midgley AC, Kong D, Li Z, Zhao Q (2019) MSC-derived sEVs enhance patency and inhibit calcification of synthetic vascular grafts by immunomodulation in a rat model of hyperlipidemia. Biomaterials 204:13–24. https://doi.org/10.1016/j.biomaterials.2019.01.049
42. Liu Y, Xue X, Zhang H, Che X, Luo J, Wang P, Xu J, Xing Z, Yuan L, Liu Y, Fu X, Su D, Sun S, Zhang H, Wu C, Yang J (2019) Neuronal-targeted TFEB rescues dysfunction of the autophagy-lysosomal pathway and alleviates ischemic injury in permanent cerebral ischemia. Autophagy 15(3):493–509. https://doi.org/10.1080/15548627.2018.1531196
43. Wang M, Wang Y, Chen Y, Gu H (2013) Improving endothelialization on 316L stainless steel through wettability controllable coating by sol–gel technology. Appl Surf Sci 268:73–78. https://doi.org/10.1016/j.apsusc.2012.11.159
44. Lee JH, Lee SJ, Khang G, Lee HB (2000) The effect of fluid shear stress on endothelial cell adhesiveness to polymer surfaces with wettability gradient. J Colloid Interface Sci 230(1):84–90. https://doi.org/10.1006/jcis.2000.7080
45. Shi J, Chen S, Wang L, Zhang X, Gao J, Jiang L, Tang D, Zhang L, Midgley A, Kong D, Wang S (2019) Rapid endothelialization and controlled smooth muscle regeneration by electrospun heparin-loaded polycaprolactone/gelatin hybrid vascular grafts. J Biomed Mater Res Part B 107(6):2040–2049. https://doi.org/10.1002/jbm.b.34295
46. Tseders ÉÉ, Purinya BA (1975) The mechanical properties of human blood vessels relative to their location. Polym Mech 11(2):271–275. https://doi.org/10.1007/BF00854734
47. Ye P, Wei S, Luo C, Wang Q, Li A, Wei F (2020) Long-term effect against methicillin-resistant staphylococcus aureus of emodin released from coaxial electrospinning nanofiber membranes with a biphasic profile. Biomolecules 10(3):362. https://doi.org/10.3390/biom10030362
48. Tort S, Han D, Steckl AJ (2020) Self-inflating floating nanofiber membranes for controlled drug delivery. Int J Pharm 579:119164. https://doi.org/10.1016/j.ijpharm.2020.119164
49. Qu B, Yuan L, Yang L, Li J, Lv H, Yang X (2019) Polyurethane end-capped by tetramethylpyrazine-nitrone for promoting endothelialization under oxidative stress. Adv Healthc Mater 8(20):1900582. https://doi.org/10.1002/adhm.201900582
50. Wang Z, Lu Y, Qin K, Wu Y, Tian Y, Wang J, Zhang J, Hou J, Cui Y, Wang K, Shen J, Xu Q, Kong D, Zhao Q (2015) Enzyme-functionalized vascular grafts catalyze in-situ release of nitric oxide from exogenous NO prodrug. J Control Release 210:179–188. https://doi.org/10.1016/j.jconrel.2015.05.283
51. Yang J, Wei K, Wang Y, Li Y, Ding N, Huo D, Wang T, Yang G, Yang M, Ju T, Zeng W, Zhu C (2018) Construction of a small-caliber tissue-engineered blood vessel using icariin-loaded β-cyclodextrin sulfate for in situ anticoagulation and endothelialization. Sci China Life Sci 61(10):1178–1188. https://doi.org/10.1007/s11427-018-9348-9
52. Guo X, Wang X, Li X, Jiang YC, Han S, Ma L, Guo H, Wang Z, Li Q (2020) Endothelial cell migration on poly(ε-caprolactone) nanofibers coated with a nanohybrid Shish–Kebab structure mimicking collagen fibrils. Biomacromol 21(3):1202–1213. https://doi.org/10.1021/acs.biomac.9b01638
53. Wang Z, Zheng W, Wu Y, Wang J, Zhang X, Wang K, Zhao Q, Kong D, Ke T, Li C (2016) Differences in the performance of PCL-based vascular grafts as abdominal aorta substitutes in healthy and diabetic rats. Biomater Sci 4(10):1485–1492. https://doi.org/10.1039/C6BM00178E
54. Xu X, Liu X, Yu L, Ma J, Yu S, Ni M (2020) Impact of intracoronary nicorandil before stent deployment in patients with acute coronary syndrome undergoing percutaneous coronary intervention. Exp Ther Med 19(1):137–146. https://doi.org/10.3892/etm.2019.8219
55. Yang HL, Korivi M, Chen CH, Peng WJ, Chen CS, Li ML, Hsu LS, Liao JW, Hseu YC (2017) Antrodia camphorata attenuates cigarette smoke-induced ROS production, DNA damage, apoptosis, and inflammation in vascular smooth muscle cells, and atherosclerosis in ApoE-deficient mice. Environ Toxicol 32(8):2070–2084. https://doi.org/10.1002/tox.22422
56. Fojta M, Daňhel A, Havran L, Vyskočil V (2016) Recent progress in electrochemical sensors and assays for DNA damage and repair. TrAC Trends Anal Chem 79(5):160–167. https://doi.org/10.1016/j.trac.2015.11.018
57. Carrizzo A, Conte Giulio M, Sommella E, Damato A, Ambrosio M, Sala M, Scala Maria C, Aquino Rita P, De Lucia M, Madonna M, Sansone F, Ostacolo C, Capunzo M, Migliarino S, Sciarretta S, Frati G, Campiglia P, Vecchione C (2019) Novel potent decameric peptide of spirulina platensis reduces blood pressure levels through a PI3K/AKT/eNOS-dependent mechanism. Hypertension 73(2):449–457. https://doi.org/10.1161/HYPERTENSIONAHA.118.11801
58. Ahmad KA, Ze H, Chen J, Khan FU, Xuezhuo C, Xu J, Qilong D (2018) The protective effects of a novel synthetic β-elemene derivative on human umbilical vein endothelial cells against oxidative stress-induced injury: involvement of antioxidation and PI3k/Akt/eNOS/NO signaling pathways. Biomed Pharmacother 106:1734–1741. https://doi.org/10.1016/j.biopha.2018.07.107
59. Wu Y, He MY, Ye JK, Ma SY, Huang W, Wei YY, Kong H, Wang H, Zeng XN, Xie WP (2017) Activation of ATP-sensitive potassium channels facilitates the function of human endothelial colony-forming cells via Ca2+/Akt/eNOS pathway. J Cell Mol Med 21(3):609–620. https://doi.org/10.1111/jcmm.13006
60. Wang X, Pan J, Liu D, Zhang M, Li X, Tian J, Liu M, Jin T, An F (2019) Nicorandil alleviates apoptosis in diabetic cardiomyopathy through PI3K/Akt pathway. J Cell Mol Med 23(8):5349–5359. https://doi.org/10.1111/jcmm.14413
61. Huang WC, Lai CL, Liang YT, Hung HC, Liou CJ (2016) Phloretin attenuates LPS-induced acute lung injury in mice via modulation of the NF-κB and MAPK pathways. Int Immunopharmacol 40:98–105. https://doi.org/10.1016/j.intimp.2016.08.035
62. Gaafar AGA, Messiha BAS, Abdelkafy AML (2018) Nicorandil and theophylline can protect experimental rats against complete Freund’s adjuvant-induced rheumatoid arthritis through modulation of JAK/STAT/RANKL signaling pathway. Eur J Pharmacol 822:177–185. https://doi.org/10.1016/j.ejphar.2018.01.009
全文链接(可点击直接下载PDF)
http://www.jzus.zju.edu.cn/oldversion/opentxt.php?doi=10.1007/s42242-020-00107-2
